Physicist: The magnetic properties of a material are governed entirely by the configuration of the electrons in that material. In metals there are two types of electrons: bound electrons and free electrons. The free electrons are free to move between atoms, and are the cause of conductivity in metals. The bound electrons are stuck to the individual atoms.
Each electron, in addition to having charge, also has a “magnetic moment” which is a fancy way of saying that it’s a tiny bar magnet. Generally the bound electrons will be paired off in opposite spin pairs. This is like putting a North-South magnet next to a South-North magnet. They almost completely cancel each other out. However, sometimes (in iron, nickel, and cobalt for example) you’ll have one or more un-paired electrons. The magnetic fields of these electrons aren’t canceled out by another, oppositely-oriented, electron. As such they lend an overall magnetic field to the atom they inhabit.
So, some metals are attracted to magnets because they are full of tinier magnets. Those tinier magnets twist about so that they align with the field of the larger magnet. However, that just pushes the question back to “Why do magnets attract each other?”.
Those free electrons aren’t completely useless. If they’re exposed to a changing magnetic field (wave your magnet around) they’ll start moving around in “eddy currents”. Those eddy currents always try to resist the changing field (“Lenz’s law” or “the universe is a stubborn jerk law”). So all conductive metals interact with magnetic fields (otherwise generators wouldn’t work), but not in the “attracted to” kind of way.
Answer gravy: “Why do magnets attract each other?” Magnetic fields, like high school students, don’t really want to exist. A magnetic field of strength B that fills up a volume V has an associated energy
It turns out that processes that release energy are usually forces. For example; when you drop an object energy is released, and it so happens that gravity is a force. Similarly, magnets will try to line up in such a way that they will cancel out each other’s fields. Less fields = less energy. So, the process of lining up to cancel out their fields decreases the energy tied up in those fields, and as such there’s a force that tries to line up the magnets.
Also: The physics behind the magnetic properties is really nasty. Nasty enough that the math can’t be done, and computer simulations can’t be trusted (generally). Here’s a map of the (experimentally found) magnetic properties on the periodic table:

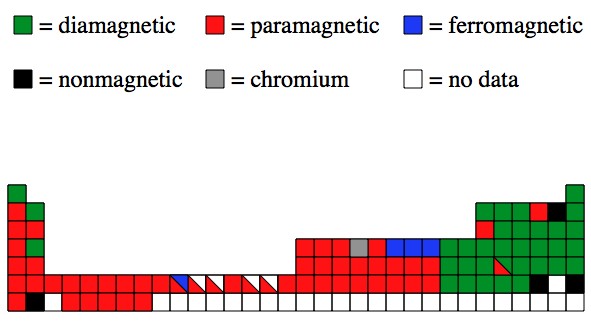






why some metals are not attracted to magnets?
please answer via email: b7a_b7a23@yahoo.com
How do any material gets color?
what type of magnet would you use to seperate cobalt from cast sands
Why does heavier iron piece attracts lighter
Magnet piece
آیا آهنربایی داریم که فلز رو دفع کند؟
okay so this is yo much reading and I have a paper do tomorrow and I need to learn about the element nickel. Thank you for your time and I will be back and I’m a newspaper writer
To which nuclei do the bonding electrons of a covalent bond revolve round ?
if we put magnet in box which is made up of lead then?
all conductive metals like cu and al are having free electron then why not they are conductive in nature?
Summy cobalt ion and nickel have parallel spins and the magnetic feild of the electrons doesn’t cancel out.
why those iron,cobalt and nickel are attracted to the magnets???
You show Al as paramagnetic, but Cu as diamagnetic. Both show Lenz effect with a strong magnet. How can that be? TIA
I have read various articles regarding why does magnet attract,but still I am not satisfied,because I yet to understand how does field created and how it attracts and repell.
Next time will say . .
Why are some materials attracted to magnets and others are not?
Very good answers
How will the magnet work, when it is used for the collection of metallic objects from the dustbin and then at particular time, the metallic objects should be released into the separate section of the dustbin (without magnetization), is it possible?
Why is that iron losses it magnetic property after some time
why is it that some metals are used to make magnets while others cannot be used. Reply via email
Why will not a magnet attract a piece of copper??
ikr
I want to know whether tungsten has magnetic properties and why some pieces of sand, granite and other stone particles are attracted towards magnets? And also I want to know why the bound electrons of an atom paired off in opposite pairs and why it does not take place in iron, nickel and cobalt like elements completely? I want an exact answer for it. You have given that iron, nickel and cobalt possess one or more unpaired electrons and because of that the magnetic fields of these electrons aren’t cancelled out by another oppositely oriented electrons. It is absolutely correct. But the metals such as Mn, Cu, Al, Cr also possess unpaired electrons know. Mn has 5 unpaired electrons, Cr has 6, 1 from ‘s’ orbital and the other 5 from ‘d’ orbital, Al and Cu each possess 1 unpaired electron. But iron has 4 unpaired electrons, nickel has 2 and cobalt has 3 unpaired electrons each. So why do the metals like Cr,Mn, Al and Cu are not attracted by magnets? They also possess unpaired electrons know? Please e-mail me and explain this to me in details. Thank you for providing me with
some valuable information about the magnetism.
The difference for non magnetic materials versus Ni, Co and Fe is that the electrons travel along orbitals that, though most are paired within the orbitals, in some cases an up spin can be countered by a down spin from an electron that is physically in another orbital yet close enough to make an impact mitigating the induction of the magnetic field. In the case of the big 3, that offsetting spin from another orbital is missing, thereby allowing just enough unpaired electrons to spin freely, inducing the magnetic moment.
i want to know why is cobalt attracted to a bar magnet
i want to know why is cobalt attracted to a bar magnet
can u harry up and tell me why cobalt attracted to a bar magnet
Unsubscribe me from these services
I like it.
what does this all mean?